Welcome, and thank you for your purchase of the LI-5800A Total Alkalinity Titrator. The LI-5800A is a total alkalinity Gran titration system that measures total alkalinity of water samples using automated controls, data logging, and calculations. If you have just taken delivery of your order, check the packaging list to be sure you received everything that you ordered.
What's what
The LI-5800A consists of the control unit, computer, pH meter, pH probe, magnetic stirrer, sample vial, and additional components.
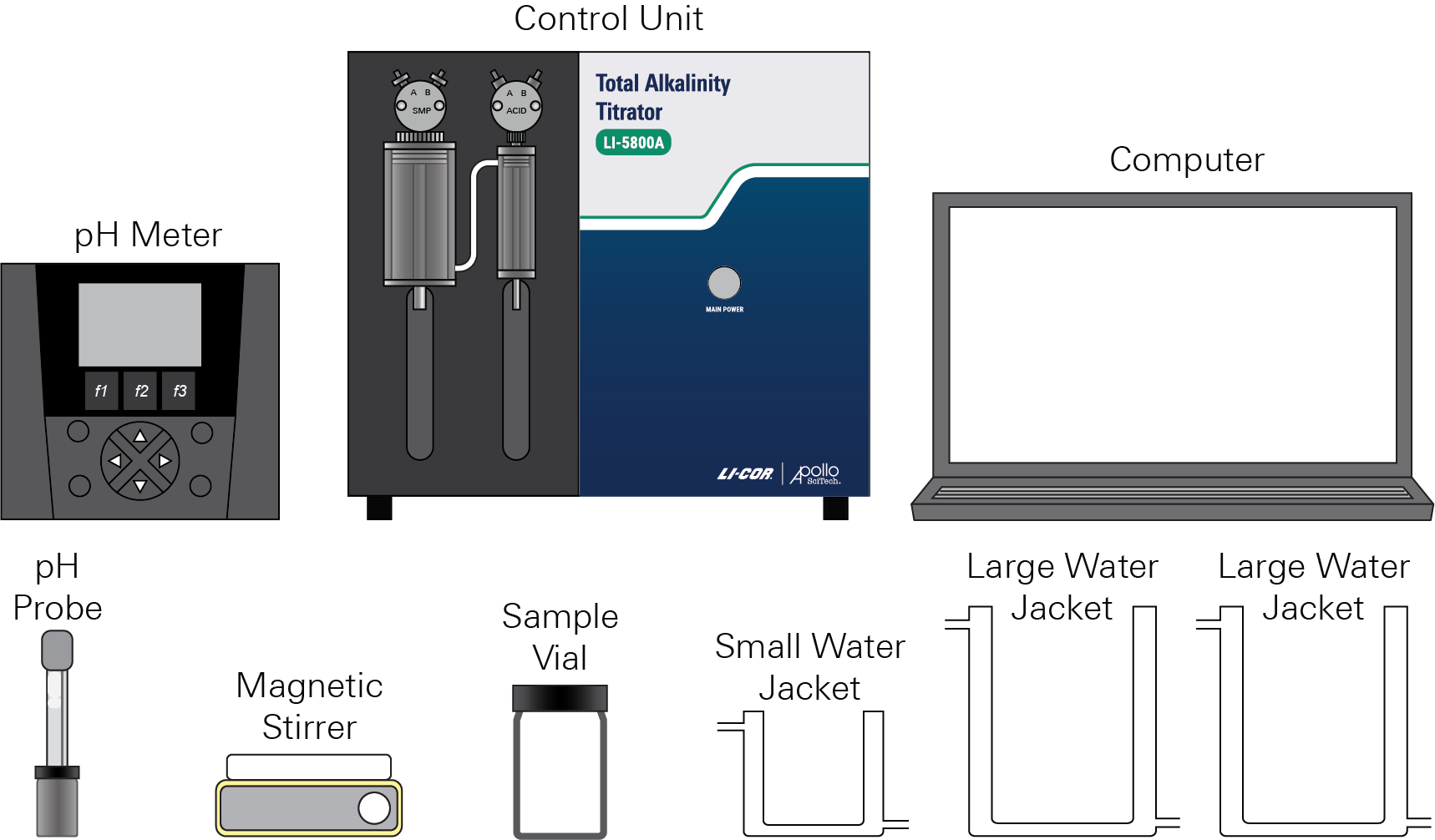
Control unit
Part number 99543-006
The control unit (Figure 1‑2) delivers samples and manages data that is stored on the computer. The titrator features two digital syringes enclosed in water baths and other analytical hardware.
The digital syringes are surrounded by water jackets to regulate the temperature of the solutions inside. For the digital syringe pumps used here, port B (input port) is on the right side and port A (output port) is on the left side.
A 3-way standard valve and a 25-mL syringe are attached to the sample/standard digital pump, which is used to transfer standard or sample solutions. A 3-way standard valve and a 1-mL syringe are attached to the titrant digital pump, which is used to transfer HCl titrant. The digital pumps are controlled by the program running on the computer.

The back of the control unit has the power cable connector, data cable connector, and a fuse.
Accessories and spares kit
Part number 99543-011
The LI-5800A includes several components, including the cables listed in Table 1‑1 and the items listed below.

Sample titration vial
Part number 300-21146
One glass vial with a lid is included. During measurements, the vial is placed in standing water in the small water jacket. It holds the stir bar and sample.
Small water jacket
Part number 99543-015
Provides thermal control of the sample in the sample titration vial. One small water jacket is included.
Large water jackets
Part number 99543-014
Provides thermal control of the acid and sample vessels. Two large water jackets are included.
pH meter (Thermo Scientific A211)
Part number 590-21255
A bench-top meter that reads the pH electrode signals. The meter includes a power adapter and a serial cable for connecting to the computer via a serial-to-USB adapter.
pH probe
Part number 590-21248
The sensor that reads pH of a sample; the pH meter is required to read data from the sensor.
Magnetic stirrer
Part number 590-21161
A magnetic stirrer is included.
Stir bar
Part number 590-21160
One stir bar is included with the device.
Computer with software
Part number 616-21277
A Dell™ Latitude™ 3450 laptop computer running Windows® 11. Includes a universal power supply and battery. The computer is equipped with a licensed version of the software required by the instruments, and should be dedicated to the control and operation of the device.
Power cable options
Each LI-5800A will include a power cable with one of the following adapters:
| Description | Part Number |
|---|---|
| For USA, Canada, Mexico, and Japan; NEMA 5-15; Type B | 390-00787 |
| For Europe, South America, and Asia; CEE 7/7; Type C | 390-01486 |
| For United Kingdom; Type G | 390-03089 |
| For Israel; SI32; Type H | 390-03059 |
| For Australia; Type I | 390-03095 |
User-supplied components
In addition to the components included with the system, some additional materials and hardware will be required.
pH buffer solutions
Three pH buffer solutions are required for the pH electrode calibration.
Reference standards
Total Alkalinity standard (Dickson CRM or an aged seawater with occasional checks against the CRM or your own Na2CO3 standard). See Alkalinity standard for more information.
Titrant
HCl titrant with a concentration of about 0.1 M. Some NaCl may be needed to adjust the ionic strength of the titrant to match that of the sample.
Extra stir bars
Although one is included with the system, it is always helpful to have some spares. We recommend cylindrical magnetic stirring bars that are 25 mm long and 6 mm in diameter (LI-COR part number 590-21160).
Extra titration vials
Several glass titration vials (similar to part number 300-21145 included with the system) will be useful. You may need up to 25 for one day of measurements. A 20 to 25 mL vessel is recommended for analysis volumes of 12 mL. A 30 mL vessel from Kimble1 is recommended for analysis volumes of 20 mL to 25 mL.
Water bath
A temperature-controlled water bath that is able to supply water at a temperature that is similar to the room temperature (at ~22 °C) to the water jackets. There should also be enough room in the water bath for several sample bottles to equilibrate with the temperature before titration.
Ring stand with clamp holder
Provide a metal stand with gripping attachment to hold the pH electrode.
Waste containers
Two 500-mL or larger waste solution containers to collect acid and sample after measurement.
Summary of the Gran titration method
Total (or titration) alkalinity (TAlk) is an important parameter used to characterize the acid-base properties of natural waters. It is defined as the deficiency of H+ or the excess base of the system with respect to the zero proton level at the CO2 equivalence point. It can also be thought of as the amount of acid (in mol/kg or mol/L) needed to titrate a water sample to the CO2 equivalence point (about pH 4.4) at which all inorganic carbon species are converted to CO2. In seawater,
TAlk = [HCO3-] + 2[CO32-] + [OH-] + [B(OH)4-] + [Si(OH)3O-] + 2[PO43-] + [HPO42-] - [H3PO4] - [H+] - [HSO4-] ≅ [HCO3-] + 2 [CO32-] + [B(OH)4-] + [HPO42-]
Note that the dominant species that appear at the CO2 equivalence point, such as CO2aq and H2PO4-, do not appear in the definition (i.e., they do not accept H+ during titration). Due to its high pK value, silicate generally doesn’t contribute much to the TAlk unless the sample has a very high pH. In anoxic environments, HS- and NH3 may also contribute to TAlk. Traditionally, TAlk is reported in units of µeq/kg or µeq/L. However, “eq” is no longer an official IUPAC unit. We suggest using µmol/kg or µmol/L, which represents the amount of HCl used during the titration. Refer to the Dickson (1981), Dickson (1992), and Cai et al. (1998) for more details on the alkalinity definition and its meaning.
Gran titration
The principle of Gran (linear) titration lies in the fact that after all the weak bases are neutralized, the concentration of strong acid in the solution should only be a dilution of the HCl added thereafter (Gran, 1952). In other words, the [H+] change and the volume of the added acid will have a linear relationship. One can use this linear relationship to precisely determine the end-point of the titration, and therefore, the TAlk of the solution being titrated.
The Gran titration essentially linearizes (see Figure 1‑5) the titration curve by means of a simple transformation function:
1‑1GF = (v + Vo)/Vo × [H+] = (v + Vo)/Vo × 10-pH
- or -
1‑2GF = (v + Vo)/Vo × 10(E-Ek)/a
where:
GF = Gran Function
v = volume of acid added to the solution in the titration vessel
Vo = original volume of the sample
pH = pH value measured after the CO2 equivalence point (~ 4.4)
E = EMF (millivolts)
a = slope of the pH electrode measured from the three buffer solutions (mV/pH unit)
Ek = an arbitrary constant.
The term (v + Vo)/Vo corrects for the volume change in [H+]. If the value of GF is calculated far from the end point, it turns out to be linear when plotted as a function of the volume of acid added (v). We recommend the pH range for seawater Gran titrations are between 3.8 and 3.0. The same pH range must be used for both standardization and sample analysis. In some estuarine waters, groundwater, and sediment pore waters that are rich with non-carbonate weak acids such as organic acids, a lower pH range may be used. Contact LI-COR for recommendations and program modifications in these cases. Please refer to the following papers for more details.
A sample seawater titration
Figure 1‑4 represents raw data and Figure 1‑5 is the chart of data taken by an early version of AS-ALK to illustrate the principle described above. This procedure is automated in the LI-5800A via the software. You do not need to perform any of it.
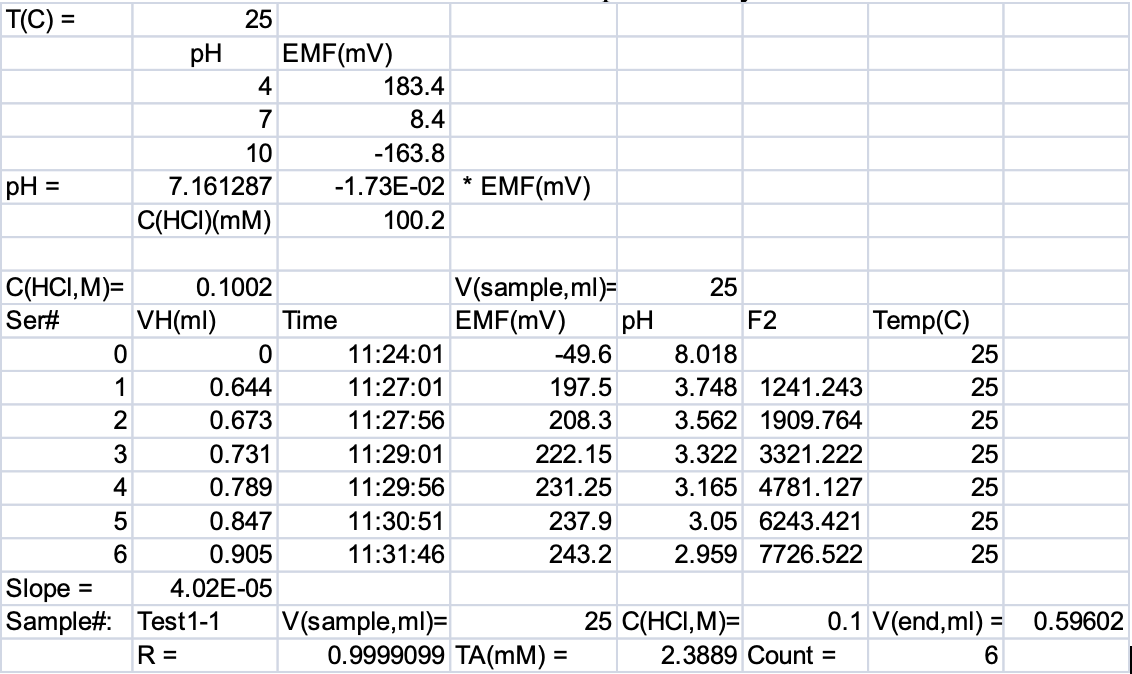

Here the end-point HCl volume is the point where GF=0. The above procedure is automated in the AS-ALK3 program. Temperature and salinity information for the sample are used to convert the TA value above into µmol/kg. Note that the choice of the best pH range within the data used to construct the Gran function is often debatable. You will find that the end-point volumes of HCl are different when different pH ranges are used. However, when standards and samples are determined in the same pH range and with the same procedures, the results are generally very reproducible.
Using temperature and salinity, the sample density can be calculated and units between mmol/L and mmol/kg can be converted using the correct equations2 in the full text of Millero and Poisson (1981).
ρ0 = 999.842594 + 6.793952×10-2T − 9.095290×10-3T2 + 1.001685×10-4T3 −
1.120083×10-6T4 + 6.536336×10-9T5
A = 8.24493×10-1 − 4.0899×10-3T + 7.6438×10-5T2 − 8.2467×10-7T3 +
5.3875×10-9T4
B = −5.72466×10-3 + 1.0227×10-4T − 1.6546×10-6T2
C = 4.8314×10-4
References
| 1 | Cai, W.-J., Wang, Y. and Hodson, R.E. (1998). Acid-Base Properties of Dissolved Organic Matter in the Estuarine Waters of Georgia, USA. 62(3), pp.473–483. doi:https://doi.org/10.1016/s0016-7037(97)00363-3. |
| 2 | Dickson, A.G. (1981). An exact definition of total alkalinity and a procedure for the estimation of alkalinity and total inorganic carbon from titration data. Deep Sea Research Part A. Oceanographic Research Papers, 28(6), pp.609–623. doi:https://doi.org/10.1016/0198-0149(81)90121-7. |
| 3 | Dickson, A.G. (1992). The development of the alkalinity concept in marine chemistry. Marine Chemistry, 40(1-2), pp.49–63. doi:https://doi.org/10.1016/0304-4203(92)90047-e. |
| 4 | Gran, G. (1952). Determination of the equivalence point in potentiometric titrations. Part II. Analyst, [online] 77(920), pp.661–671. doi:https://doi.org/10.1039/AN9527700661. |
| 5 | Millero, F.J. and Poisson, A. (1981). International one-atmosphere equation of state of seawater. Deep Sea Research Part A. Oceanographic Research Papers, 28(6), pp. 625–629. https://doi.org/10.1016/0198-0149(81)90122-9. |
